Bangladesh in no hurry to trial Sinovac's virus vaccine
Bangladesh is in no hurry to start a late-stage human trial of a vaccine for the novel coronavirus, developed by China’s Sinovac Biotech Ltd, according to the country's top health officials
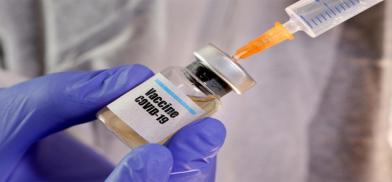
Bangladesh is in no hurry to start a late-stage human trial of a vaccine for the novel coronavirus, developed by China’s Sinovac Biotech Ltd, according to the country's top health officials.
The Bangladesh Medical Research Council or BMRC approved the phase 3 trial of Sinovac's vaccine candidate, known as CoronaVac.
But due to the novelty of the process, health authorities are awaiting approval from the government's top brass before flagging off the trials.
The International Centre for Diarrhoeal Disease Research, Bangladesh or icddr,b is set to conduct the trial of the Chinese vaccine. The vaccine will likely be administered to health workers in the country over the next 18 months.
As the trial will be held on a national level, the Ministry of Health wants to proceed with caution, Dr ABM Khurshid Alam, director-general of DGHS, told bdnews24.com. The agency is waiting for results of the trials conducted in China, he said.
“The tests will be conducted on a national level. Who will be held accountable if any harm comes from it? Although the BMRC has approved the trial, we can’t start it right away. We should take time.”
The health directorate is waiting for the government’s decision as they need the green light from the National Health Committee, BMRC, the health ministry and even the prime minister.
https://bdnews24.com/bangladesh/2020/08/12/bangladesh-in-no-hurry-to-trial-sinovac-s-virus-vaccine




Post a Comment