Dr Reddy's partners with BIRAC for Sputnik V trials in India
Indian pharma major Dr Reddy's said it has entered into a partnership with Biotechnology Industry Research Assistance Council (BIRAC) set up by the Department of Biotechnology (DBT) for advisory support on clinical trials of Russian Covid-19 vaccine Sputnik V in India
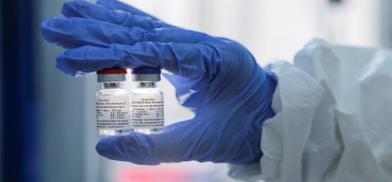
Indian pharma major Dr Reddy's said it has entered into a partnership with Biotechnology Industry Research Assistance Council (BIRAC) set up by the Department of Biotechnology (DBT) for advisory support on clinical trials of Russian Covid-19 vaccine Sputnik V in India.
The partnership will allow Dr Reddy's to identify and use some of BIRAC's clinical trial centres for the vaccine, which are funded under the National Biopharma Mission (NBM), implemented by the Project Management Unit-NBM at BIRAC. The company will also have access to Good Clinical Laboratory Practice (GCLP) labs to conduct immunogenicity assay testing of the vaccine.
"We are pleased with the collaboration with BIRAC as an advisory partner for clinical trials of the Sputnik V vaccine in India. We look forward to working with them to accelerate our efforts in bringing the vaccine to India," Satish Reddy, Chairman, Dr Reddy's Laboratories, said in a statement.
Earlier this month, Dr Reddy's and Russia Direct Investment Fund (RDIF) received approval from the Drugs Controller General of India (DCGI) to conduct an adaptive phase 2/3 human clinical trial for the Sputnik V vaccine in India.
"The government is committed to fast track clinical development of COVID vaccine candidates and provide facilitation to accelerate market readiness of a suitable vaccine," said Renu Swarup, Secretary, DBT and Chairperson, BIRAC.
On August 11, the Sputnik V vaccine developed by the Gamaleya National Research Institute of Epidemiology and Microbiology was registered by the Ministry of Health of Russia and became the world's first registered vaccine against Covid-19 based on the human adenoviral vector platform.
Opportunity for India as Russia ready to produce Covid vax at foreign sites of partners
India has a huge opportunity to be the next big hub for vaccine production after Russia announced that it will manufacture Covid-19 vaccines at foreign sites with partners.
Amir Ullah Khan, economist at MCRHRDI of the Government of Telegana, and former senior advisor for Bill and Melinda Gates Foundation, said, "As Russia is keen to work with other countries, including India, this is a huge opportunity for us to be the next big hub for vaccine production. India has played an important role with the most prominent partnership of Dr. Reddy's lab with the Russian Development Investment Fund. The lab will conduct clinical trials and provide Sputnik V for coronavirus in India."
Russia has recently expressed the willingness to produce vaccines at foreign sites. It is very encouraging and a huge opportunity for India which is among the largest manufacturers of generic drugs and vaccines in the world. It is home to half a dozen major vaccine makers which are developing vaccines against the virus that causes Covid-19.
"With India's increasing experience in manufacturing vaccines and crucial role in product innovation that is affordable, the country is foreseen as a growing vaccine hub. About 60 per cent of all vaccines worldwide are manufactured in India and it can be world's door to vaccines," said Gajendra Singh, a public health expert.
(IANS)





Post a Comment