Russia to produce Sputnik V Covid vaccine in India
The Russian Direct Investment Fund (RDIF) and Hetero, one of India's generic pharmaceutical companies, have agreed to produce in India over 100 million doses per year of Sputnik V COVID-19 vaccine in India
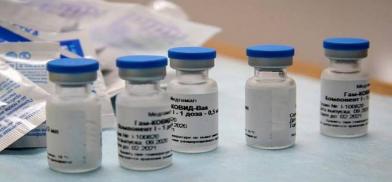
The Russian Direct Investment Fund (RDIF) and Hetero, one of India's generic pharmaceutical companies, have agreed to produce in India over 100 million doses per year of Sputnik V COVID-19 vaccine in India.
The parties intend to start the production of Sputnik V at the beginning of 2021.
"We are pleased to collaborate with RDIF as a manufacturing partner for the most anticipated Sputnik V vaccine for the treatment of COVID-19," Murali Krishna Reddy, Director, International Marketing, Hetero Labs Limited said in a statement.
"This collaboration is another step towards our commitment in the battle against COVID-19 and realizing the objective of 'Make-in-India' campaign as envisioned by our Prime Minister of India," Reddy added.
According to the RDIF -- Russia's sovereign wealth fund, the vaccine's interim clinical trial results show 95 per cent efficacy on the 42nd day after the first dose.
Currently, Phase III clinical trials are approved and are ongoing in Belarus, the UAE, Venezuela and other countries, as well as Phase II-III in India.
Requests for more than 1.2 billion doses of Sputnik V vaccine came from more than 50 countries.
The vaccine supplies for the global market will be produced by RDIF's international partners in India, Brazil, China, South Korea and other countries.
"We are delighted to announce the agreement between RDIF and Hetero that will pave the way to the production of the safe and highly effective Sputnik V vaccine on Indian soil," Kirill Dmitriev, CEO of the Russian Direct Investment Fund, said in a statement.
"Thanks to our cooperation with Hetero, we will be able to significantly increase production capacity and provide people of India with an efficient solution in this challenging period of the pandemic," Dmitriev added.
The Russian vaccine uses two different vectors based on the human adenovirus, rather than using one and the same vector for two doses.
(IANS)





Post a Comment